Home
Simulation Example
Article (280kB
PDF)
Downloads
Working Group
Bill
Boettinger
Ursula
Kattner
Kil-Won Moon
|
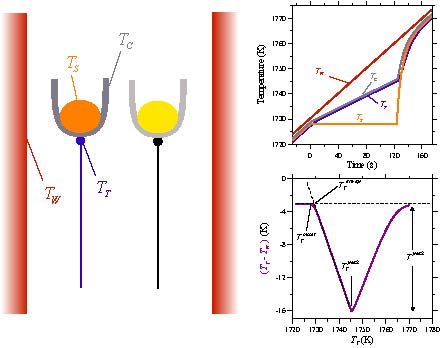
Differential thermal analysis (DTA) measurements are a standard method
of determining standard method of determining transformation temperatures
of materials. Accurate interpretation of these measurements is essential
when they are used to infer the location of equilibrium phase diagram boundaries.
The accuracy of the measurements is affected by the details of heat transfer
within the measurement cell and by transformation kinetics. Simulations
can provide understanding into how thermal events from transformations are
reflected in the DTA signal and can enable more accurate interpretation of
DTA results from samples with unknown characteristics. A heat flow model
of a DTA and thermal characteristics obtained by fitting experimental results
for a pure metal, the response of the DTA is modeled for the melting
and solidification of alloys.
As input to the calculation we use enthalpy vs. temperature values
obtained for full equilibrium (lever law)
and for Scheil freezing assumptions.
It should be noted that these are extreme cases. DTA melting simulations,
using the lever enthalpy-temperature relation, would apply to an alloy
equilibrated prior to melting and where diffusion was adequate to guarantee
spatial concentration uniformity of all phases during melting. DTA melting
simulations, using the Scheil enthalpy calculations, would apply to a microstructure
that was solidified and remelted with no solid diffusion. The melting of
an equilibrated alloy as well as an as-cast sample requires an analysis
of solid diffusion for both the freezing process and the melting process.
|

